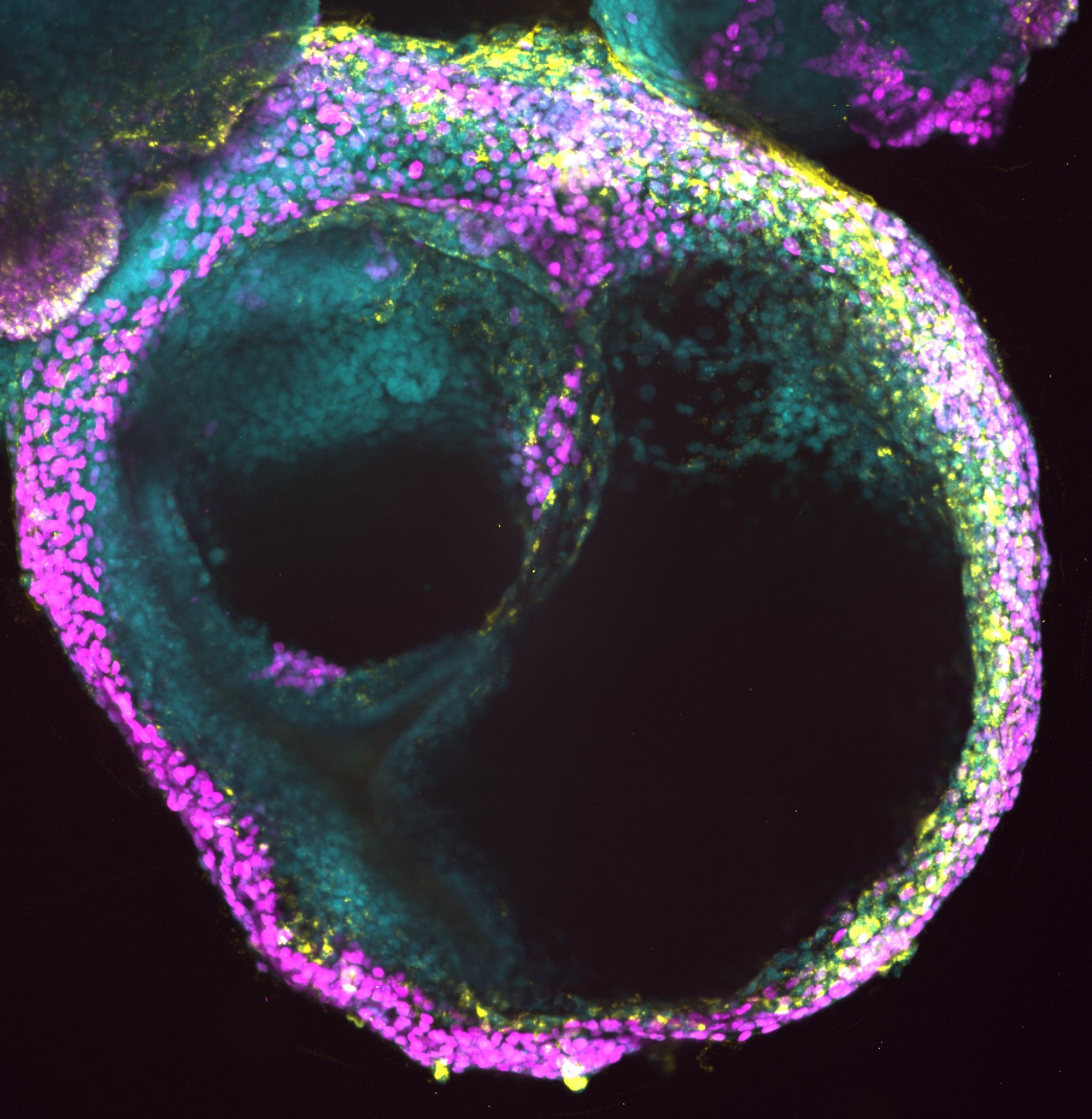
Derivation of Human HSPCs from Hemogenic Gastruloids
Summary
Hematopoietic stem and progenitor cells (HSPCs) are the precursors of all blood and immune cell types. Their derivation from pluripotent stem cells (PSCs) in vitro has long been a central goal of regenerative medicine, but true HSC-like cells capable of long-term multi-lineage reconstitution have remained elusive. A key reason is that conventional embryoid body (EB)-based differentiation protocols produce largely unstructured aggregates that rely on stepwise external addition of known cytokines and growth factors. These approaches typically bias cells toward a single lineage with limited stromal support and require laborious re-optimization for each PSC line. Most critically, they fail to reconstitute the aorta-gonad-mesonephros (AGM) niche — the multi-tissue embryonic environment where definitive HSCs naturally emerge through endothelial-to-hematopoietic transition (EHT).
In the embryo, HSC specification is not driven by hemogenic endothelium in isolation. The AGM is a multi-tissue structure in which the hemogenic endothelium receives spatially and temporally patterned inputs from surrounding tissues: paraxial mesoderm- and somite-derived Wnt16 and Notch ligands instruct arterial identity of the hemogenic endothelium; intermediate mesoderm provides SCF and retinoic acid supporting HSC maturation; subaortic mesenchyme (PDGFRα⁺ cells of mesodermal origin) delivers direct niche support and shapes the BMP microenvironment permissive for EHT; and gut epithelium provides instructive Hedgehog signals responsible for the ventral polarity of intra-aortic cluster formation. No EB-based protocol recapitulates this multi-tissue architecture — the signals it provides arrive externally, uniformly, and in sequence, rather than emerging from within an organized tissue context.
Our approach: self-organized AGM niche reconstitution
Our hemogenic gastruloid project takes a fundamentally different strategy: we exploit the intrinsic self-organizing capacity of pluripotent stem cells to generate a multi-tissue environment internally, rather than reconstructing it signal by signal from the outside. By providing a small set of global developmental cues, we direct human embryonic stem cells (hESCs) and induced pluripotent stem cells (hiPSCs) to form hemogenic gastruloids — three-dimensional, self-organizing structures that spontaneously produce key components of the AGM niche alongside the hemogenic endothelial lineage itself. Proof of concept that gastruloid systems can capture hematopoietic development with spatiotemporal fidelity has been established in mouse models (Rossi et al., Sci. Rep. 2022; Ragusa et al., eLife 2025), but these studies used mouse embryonic stem cells and captured only early hematopoietic waves. We extend this approach to human cells, where HSC specification involves distinct gene regulatory logic and niche dependencies.
What we have established
Using unbiased single-cell RNA sequencing, we find that human hemogenic gastruloids spontaneously generate gut epithelium, subaortic mesenchyme, and associated stromal populations alongside the hemogenic endothelial lineage — demonstrating partial, self-organized reconstitution of the AGM niche without external instruction of each component. Within this environment, cells undergo EHT and produce CD34⁺CD38⁻CD90⁺CD45RA⁻ HSPCs whose transcriptional profiles co-embed with human AGM-stage definitive HSCs from in vivo reference atlases (Calvanese et al., Nature 2022), and which generate erythroid, granulocyte-macrophage, and multipotent colonies as well as CD5⁺CD7⁺ T-lymphoid progenitors — confirming definitive hematopoietic identity.
Approach and long-term goal
We are currently working to improve and mechanistically understand this system using single-cell RNA sequencing combined with expressed DNA barcoding to map which hemogenic endothelial subpopulations most reliably give rise to high-quality HSPCs, and to reconstruct state-to-fate relationships across the EHT transition; and spatio-temporal imaging including HCR FISH, whole-mount immunostaining, and quantitative live imaging to resolve the spatial organization of hemogenic and niche cell types within intact gastruloids. In parallel, we are systematically modifying culture conditions to expand paraxial and intermediate mesoderm compartments, aiming to more fully reconstitute the AGM signaling environment and improve HSPC yield and quality. The long-term goal is to derive clinically relevant human HSCs capable of long-term multi-lineage reconstitution — a milestone that would enable patient-specific, transplantable blood stem cell therapies independent of matched donors.
Key references
Calvanese V, Capellera-Garcia S, Ma F, et al. Mapping human haematopoietic stem cells from haemogenic endothelium to birth. Nature. 2022;604:534–540. doi:10.1038/s41586-022-04571-x
Rossi G, Giger S, Hübscher T, Lutolf MP. Gastruloids as in vitro models of embryonic blood development with spatial and temporal resolution. Sci. Rep. 2022;12:13380. doi:10.1038/s41598-022-17265-1
Ragusa D, Suen C-W, Torregrosa-Cortés G, et al. Dissecting infant leukemia developmental origins with a hemogenic gastruloid model. eLife. 2025;13:e102324. doi:10.7554/eLife.102324


